Depth of heart, depth of care – April 2026
Advancing Cardiac Care
As adult congenital heart disease (ACHD) populations continue to grow, clinical teams face a familiar challenge: delivering accurate right ventricular (RV) functional assessment consistently, without placing unsustainable demands on MRI services.
This month, we highlight new ACHD evidence supporting VMS+ core technology, three-dimensional knowledge-based reconstruction (3D-KBR), and reflect on how advanced RV imaging can be better aligned with real-world clinical workflows.
The PLUS of VMS+
RV Surveillance in Adults with Repaired Tetralogy of Fallot
Accurate assessment of RV volume and function is central to the long-term management of adults with repaired Tetralogy of Fallot (rToF). While cardiac MRI (cMRI) remains the gold standard, repeated access is not always feasible for routine surveillance.
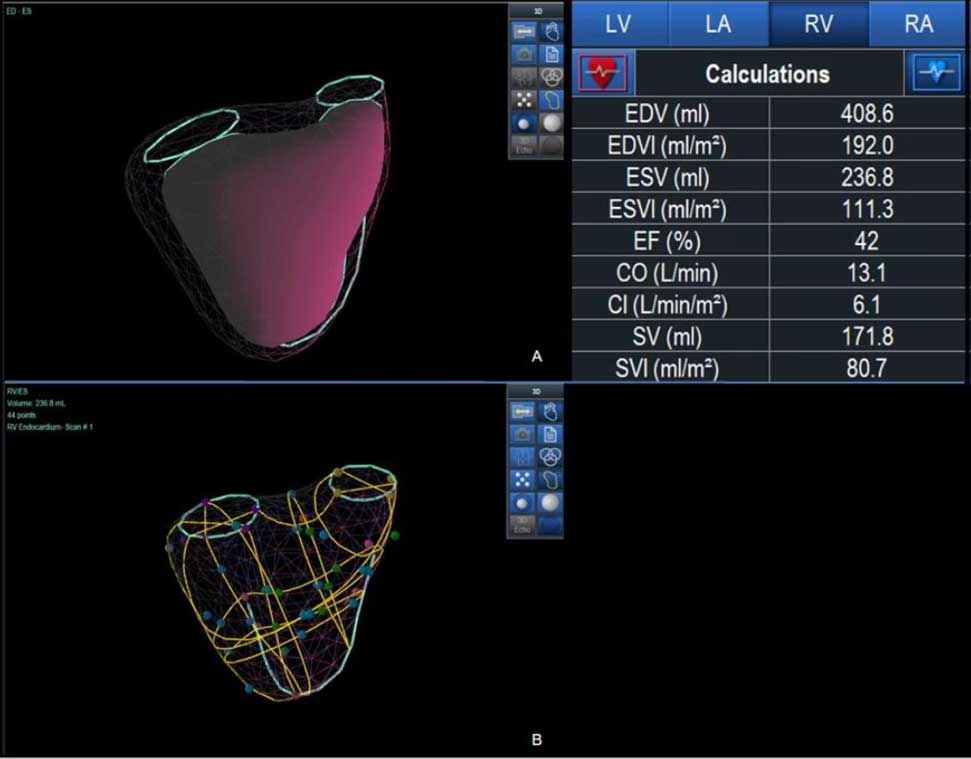
At the British Society of Echocardiography (BSE) 2025 conference, Dario Freitas and colleagues from Guy’s & St Thomas’ NHS Foundation Trust presented prospective data evaluating 3D-KBR (VMS+ core technology) for RV assessment in ACHD patients.
Study overview
- 34 adults with repaired ToF and moderate–severe pulmonary regurgitation
- 22 healthy controls
- VMS+ using standard 2D echo compared to cMRI
- Intra- and inter-observer reproducibility assessed
Key findings
- High feasibility (>85% successful acquisitions)
- Excellent reproducibility between operators
- Strong correlation with cMRI for RV volumes
- Small underestimation of EF, remaining within 10% of cMRI values
“Although cardiac MRI remains the gold standard, our findings suggest that three-dimensional knowledge-based reconstruction is a feasible and reproducible technique for longitudinal assessment of the right ventricle in adults with repaired Tetralogy of Fallot.”
— Dario Freitas, Clinical Scientist and Lead for ACHD Echocardiography,
Guy’s & St Thomas’ NHS Foundation Trust
Why This Matters
Earlier detection of progressive RV dilatation can support timely clinical decisions and reduce reliance on a single imaging modality for interim follow-up.
Tech+
Making Advanced RV Imaging Work in Everyday CHD Care
Each month, Tech+ looks at how technology fits into clinical reality. This month’s focus: workflow.
Across recent CHD discussions, including our North American webinar, one message was consistent — the barrier is not understanding best practice, but delivering it reliably.
“The challenge for many CHD services isn’t knowing what optimal imaging looks like — it’s delivering that level of assessment consistently, within real clinical constraints.”
CHD teams frequently face:
- Limited access to cMRI for routine follow-up
- Growing patient volumes requiring lifelong surveillance
- Variation in measurements across operators and timepoints
- Advanced echocardiographic tools gain traction when they:
- Fit existing echo protocols
- Support reproducible, quantitative outputs
- Enable consistent longitudinal comparison
Rather than replacing cMRI, echo-based 3D reconstruction supports a layered imaging strategy — combining MRI for major decision points with echo-based volumetric assessment for interim surveillance.

VMS+ clinical workflow
Community Spotlight
Supporting CHD Teams Beyond the Scan
Our clinical and research partners continue to shape how VMS+ evolves — from ACHD validation studies to practical discussions around scalability and workflow integration. Your insight drives how we support sustainable, high-quality cardiac imaging.
Join the VMS+ Movement
Looking to enhance RV surveillance without increasing clinical burden?
- Request a Demo – see how VMS+ integrates into your echo workflow
- Share Your Experience – tell us how your team approaches RV assessment
- Connect With Us – your feedback helps shape our roadmap
Let’s transform cardiac care together:
one patient, one insight, one workflow at a time.
Warm regards,
Matt Dobson,
Marketing Director
Ventripoint Diagnostics Ltd
VMS+ is for trained Healthcare Professional use only.
Availability subject to regulatory approval in respective regions: please contact Ventripoint Diagnostics Ltd. for further information.

